Our Research Focuses on Engineering Phosphorus-Containing Molecules that are useful as (I) Leads for Medicinal Chemistry and/or (II) Tools for Asymmetric Synthesis.
There are obvious benefits to combining studies in novel synthetic methodologies with studies in medicinal chemistry. Under the large umbrella of our two research themes (I and II), several of our projects represent unique examples of the extensive “cross-pollination” between our efforts in medicinal chemistry (I) and those in catalytic and asymmetric synthesis (II).
1. Fraser G. Ferens, Daniel D. Waller, Rebecca Boutin, Hiu-Fung Lee, Marc Saba El Leil, Mathieu Tremblay, Tian Lai Guan, Kathryn Skorey, Michael Sebag, Arun P. Wiita, M. Joanne Lemieux, Youla S. Tsantrizos.
Structural and Functional Analysis of GGPPS Inhibition as a Therapeutic Mechanism for Acute Myeloid Leukemia Pre Print 2025
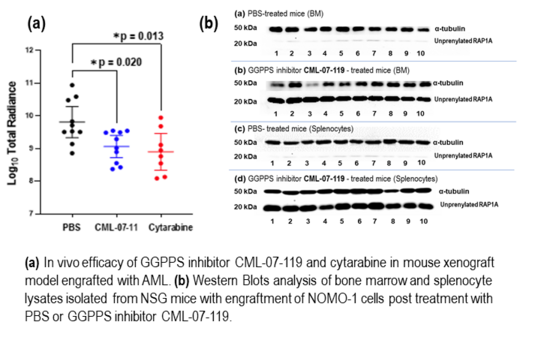 Read PDF...
Read PDF...
2. Taeok Kim, Fuqing Jin, Hatem M. Titi, and Youla S. Tsantrizos.
Diastereoselective Synthesis of Phosphinyl Peptides via Rh-Catalyzed 1,4-Addition in Coparticipation of a P-Chiral Moiety and Difluorphos. J. Org. Chem. 2024
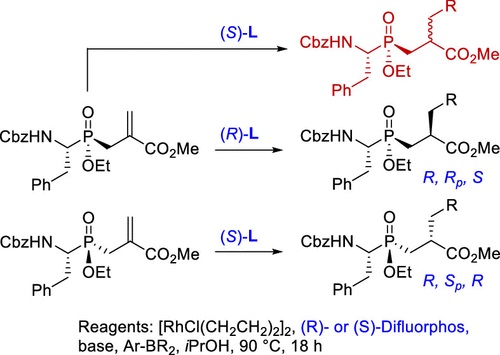 Read PDF...
Read PDF...
3. Ifenna Mbaezue, Shi-Guang Li, Angula C. S. Reddy, Hatem M. Titi, and Youla S. Tsantrizos.
Solvent-Switchable Remote C–H Activation via 1,4-Palladium Migration Enables Site-Selective C–P Bond Formation: A Tool for the Synthesis of P-Chiral Phosphinyl Imidazoles. Org. Lett. 2024
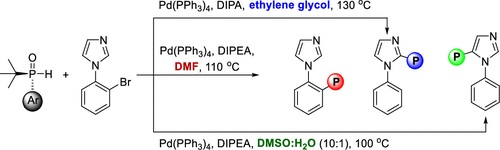 Read PDF...
Read PDF...
4. Rebecca Boutin, Hiu-Fung Lee, Tian Lai Guan, Tan Trieu Nguyen, Xian Fang Huang, Daniel D. Waller, Jordan Lu, Iok In Christine Chio, René P. Michel, Michael Sebag, and Youla S. Tsantrizos.
Discovery and Evaluation of C6-Substituted Pyrazolopyrimidine Based Bisphosphonate Inhibitors of the Human Geranylgeranyl Pyrophosphate Synthase and Evaluation of Their Antitumor Efficacy in Multiple Myeloma, Pancreatic Ductal Adenocarcinoma, and Colorectal Cancer. J. Med. Chem. 2023
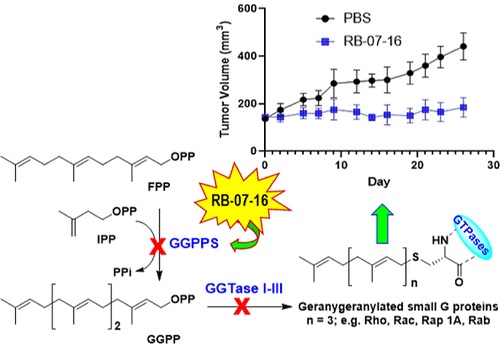 Read PDF...
Read PDF...
5. Ifenna I. Mbaezue, Filip Topic and Youla S. Tsantrizos.
Re-evaluation of P-Chiral, N-Phosphoryl Sulfonamide Brønsted Acids in the Asymmetric Synthesis of 1,2,3,4-Tetrahydroquinoline-2-carboxylate Esters via Biomimetic Transfer Hydrogenation. Synlett 2023
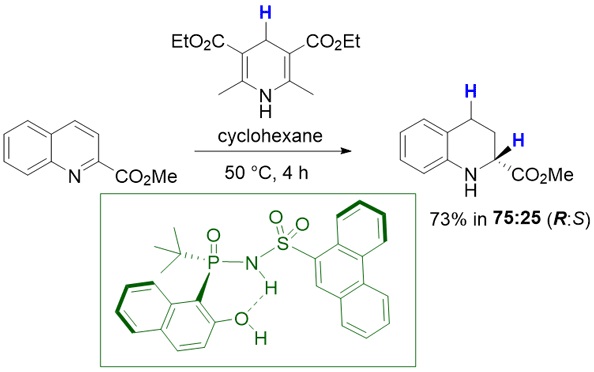 Read PDF...
Read PDF...
Our research is multidisciplinary, involving a seamless integration of fundamental and applied sciences, and provides an excellent training ground for students and Post-Doctoral Fellows interested in pursuing an academic or industrial career.
